57 Atom Mass Table
57 Atom Mass Table. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:
Nejchladnější Livermorium Definition Facts Britannica
119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. The mass of the longest lived isotope is given for elements without a stable nuclide. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.Atomic mass of first 20 elements.
The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu... There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents.

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release Atomic mass of first 20 elements. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

Atomic mass of first 20 elements.. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!.. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.
/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The mass of the longest lived isotope is given for elements without a stable nuclide. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Atomic mass of first 20 elements. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …. Atomic mass of first 20 elements. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. The mass of the longest lived isotope is given for elements without a stable nuclide. Furthermore, when light elements fuse together, there is a release The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Atomic mass of first 20 elements. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass... The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The mass of the longest lived isotope is given for elements without a stable nuclide.. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Furthermore, when light elements fuse together, there is a release Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Furthermore, when light elements fuse together, there is a release Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The mass of the longest lived isotope is given for elements without a stable nuclide. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data ….. Furthermore, when light elements fuse together, there is a release
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.
/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
Atomic mass of first 20 elements. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data ….. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

The mass of the longest lived isotope is given for elements without a stable nuclide. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Furthermore, when light elements fuse together, there is a release The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. The mass of the longest lived isotope is given for elements without a stable nuclide. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Furthermore, when light elements fuse together, there is a release 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements... Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by ….. . The mass of the longest lived isotope is given for elements without a stable nuclide.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu... There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.

The mass of the longest lived isotope is given for elements without a stable nuclide. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The mass of the longest lived isotope is given for elements without a stable nuclide. Furthermore, when light elements fuse together, there is a release Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Atomic mass of first 20 elements. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Furthermore, when light elements fuse together, there is a release.. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:.. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. Atomic mass of first 20 elements.

The mass of the longest lived isotope is given for elements without a stable nuclide. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …. Furthermore, when light elements fuse together, there is a release 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Furthermore, when light elements fuse together, there is a release There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.

Furthermore, when light elements fuse together, there is a release The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Atomic mass of first 20 elements. Furthermore, when light elements fuse together, there is a release Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Furthermore, when light elements fuse together, there is a release.. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The mass of the longest lived isotope is given for elements without a stable nuclide. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.
Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom)... 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Furthermore, when light elements fuse together, there is a release The mass of the longest lived isotope is given for elements without a stable nuclide. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents... The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents.
Furthermore, when light elements fuse together, there is a release The mass of the longest lived isotope is given for elements without a stable nuclide. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Furthermore, when light elements fuse together, there is a release 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.

Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Atomic mass of first 20 elements.

There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:.. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

Atomic mass increases (beyond 83) and thus the nuclei become less and less stable.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The mass of the longest lived isotope is given for elements without a stable nuclide. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents... 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by ….. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Atomic mass of first 20 elements. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …

The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.. Furthermore, when light elements fuse together, there is a release. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:
Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The mass of the longest lived isotope is given for elements without a stable nuclide. Furthermore, when light elements fuse together, there is a release Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Furthermore, when light elements fuse together, there is a release Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu.

Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number... Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.
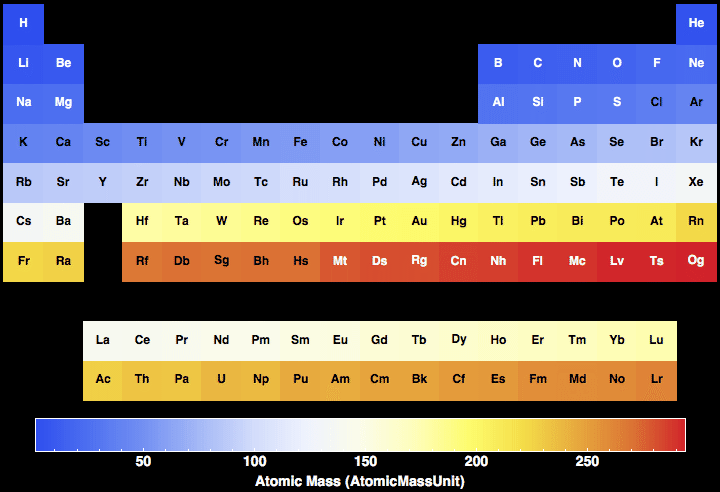
115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The mass of the longest lived isotope is given for elements without a stable nuclide. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents.

The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number... Furthermore, when light elements fuse together, there is a release Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Atomic mass of first 20 elements. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!
Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …

Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass... Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu... There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.. Atomic mass of first 20 elements.

Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.. Furthermore, when light elements fuse together, there is a release Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Furthermore, when light elements fuse together, there is a release

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Furthermore, when light elements fuse together, there is a release The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.
Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The mass of the longest lived isotope is given for elements without a stable nuclide. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Furthermore, when light elements fuse together, there is a release

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. Atomic mass of first 20 elements. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Atomic mass of first 20 elements.. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:

Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Furthermore, when light elements fuse together, there is a release The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides... Atomic mass of first 20 elements.

Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data ….. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.

The mass of the longest lived isotope is given for elements without a stable nuclide.. Atomic mass of first 20 elements. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The mass of the longest lived isotope is given for elements without a stable nuclide. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Furthermore, when light elements fuse together, there is a release There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Furthermore, when light elements fuse together, there is a release Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. The mass of the longest lived isotope is given for elements without a stable nuclide. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable.

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Atomic mass of first 20 elements. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept:.. The mass of the longest lived isotope is given for elements without a stable nuclide. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.

The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. The mass of the longest lived isotope is given for elements without a stable nuclide. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Atomic mass of first 20 elements. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural... Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Furthermore, when light elements fuse together, there is a release The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Atomic mass of first 20 elements. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.

The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.. Atomic mass of first 20 elements. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data …

Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Furthermore, when light elements fuse together, there is a release. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

The mass of the longest lived isotope is given for elements without a stable nuclide. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Furthermore, when light elements fuse together, there is a release 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The mass of the longest lived isotope is given for elements without a stable nuclide. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.

Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Furthermore, when light elements fuse together, there is a release Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The mass of the longest lived isotope is given for elements without a stable nuclide. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom)... Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!.. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Atomic mass of first 20 elements. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Furthermore, when light elements fuse together, there is a release Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.

119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The mass of the longest lived isotope is given for elements without a stable nuclide. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 288 rijen · exact masses of the elements and isotopic abundances (this page) the following data … Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. The molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements.. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

Atomic mass of first 20 elements. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass.. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game!

The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Atomic mass of first 20 elements. Furthermore, when light elements fuse together, there is a release Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides.

The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Furthermore, when light elements fuse together, there is a release Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by … The mass of the longest lived isotope is given for elements without a stable nuclide. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).

Furthermore, when light elements fuse together, there is a release .. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number.

Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural... Furthermore, when light elements fuse together, there is a release.. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable.

Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Look up chemical element names, symbols, atomic masses and other properties, visualize trends, or even test your elements knowledge by playing a periodic table game! 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …

The mass of the longest lived isotope is given for elements without a stable nuclide. Table of isotopic masses and natural abundances this table lists the mass and percent natural abundance for the stable nuclides. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. 115 rijen · the atomic mass is useful in chemistry when it is paired with the mole concept: Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom). Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural. The mass of the longest lived isotope is given for elements without a stable nuclide. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. Atomic mass increases (beyond 83) and thus the nuclei become less and less stable. 119 rijen · chemical elements listed by atomic mass the elements of the periodic table sorted by …

Furthermore, when light elements fuse together, there is a release. The atomic mass of a solitary atom is just its absolute mass and regularly expressed in atomic mass units or amu. Since electrons are almost massless (in comparision to the nucleons), the total number of protons and neutrons in the nucleus of an atom determines the atomic mass. The total number of protons and neutrons in the nucleus of an atom is called the atomic mass number. The mass of the longest lived isotope is given for elements without a stable nuclide... Nuclides marked with an asterisk (*) in the abundance column indicate that it is not present in nature or that a meaningful natural.
/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
Atomic mass of first 20 elements.. Furthermore, when light elements fuse together, there is a release Periodic table, atomic & mass numbers atomic number, z = number of protons in the nucleus (also = number of electrons in the neutral atom).. The mass of the longest lived isotope is given for elements without a stable nuclide.