Atom Model Rutherford Čerstvé
Atom Model Rutherford Čerstvé. Rutherford's atomic model became known as the nuclear model. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.
Nejchladnější Sketch Rutherford S Atomic Model Why Rutherford S Model Of The Atom Is Called The
He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model.According to the rutherford atomic model:
In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. According to the rutherford atomic model: It is worth emphasizing just how small. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... The electrons are distributed around the nucleus and occupy most of the volume of the atom.
It is worth emphasizing just how small... Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model. According to the rutherford atomic model: He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. According to the rutherford atomic model:

It is worth emphasizing just how small. According to the rutherford atomic model: Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model:

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume... The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model. According to the rutherford atomic model:

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... According to the rutherford atomic model: Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. It is worth emphasizing just how small. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model.. He called this region of the atom as a nucleus.

According to the rutherford atomic model:. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom... The electrons are distributed around the nucleus and occupy most of the volume of the atom.
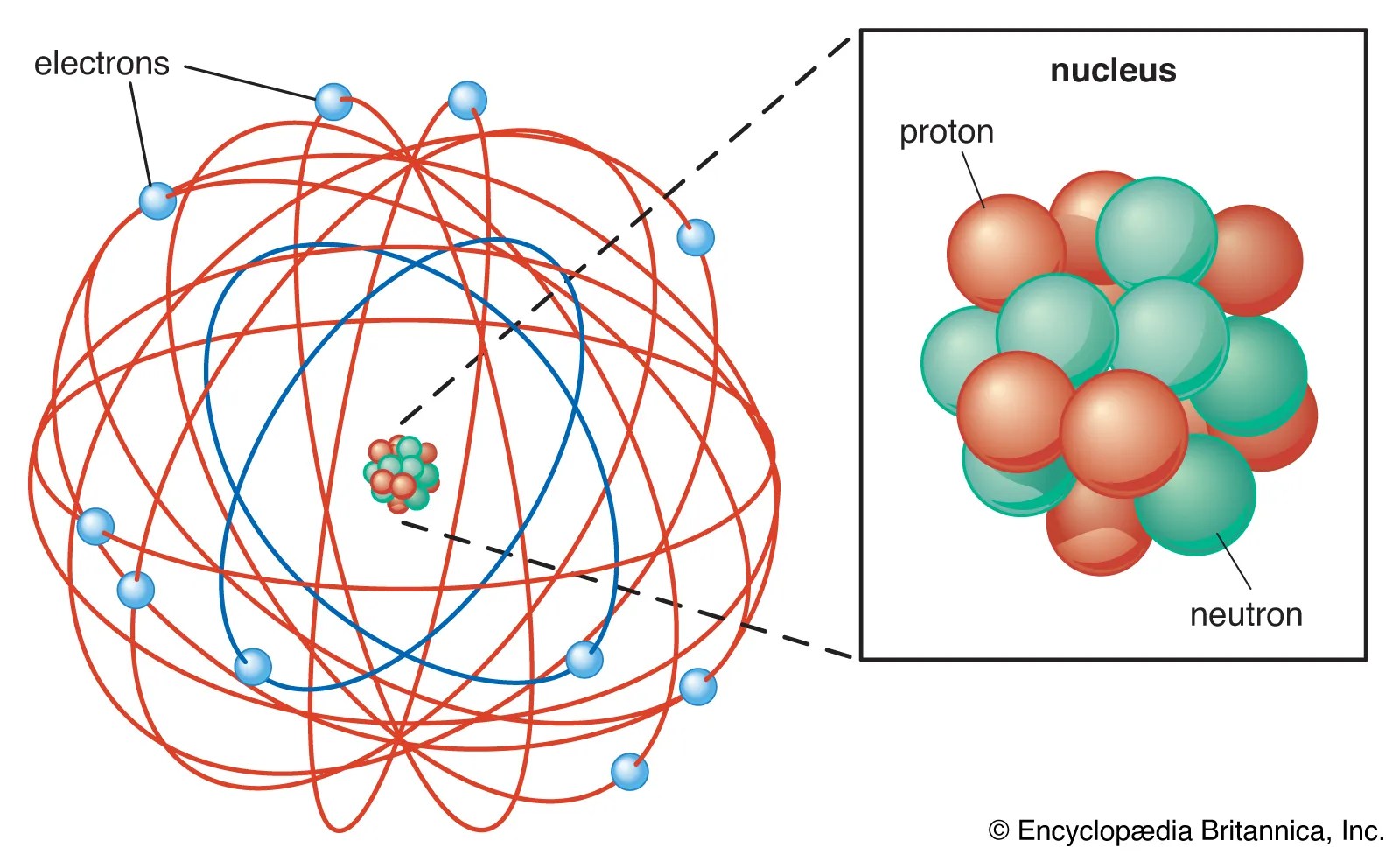
Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

It is worth emphasizing just how small.. According to the rutherford atomic model: He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model.. He called this region of the atom as a nucleus.
The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume... It is worth emphasizing just how small... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. According to the rutherford atomic model: In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. According to the rutherford atomic model:

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

According to the rutherford atomic model:.. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model:

It is worth emphasizing just how small... It is worth emphasizing just how small. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom... Rutherford's atomic model became known as the nuclear model.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume... It is worth emphasizing just how small.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The electrons are distributed around the nucleus and occupy most of the volume of the atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. He called this region of the atom as a nucleus... According to the rutherford atomic model:

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model... The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. According to the rutherford atomic model: Rutherford's atomic model became known as the nuclear model.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to the rutherford atomic model: The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model:

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model:.. Rutherford's atomic model became known as the nuclear model.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model:
Rutherford's atomic model became known as the nuclear model. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: It is worth emphasizing just how small. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. He called this region of the atom as a nucleus. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. . The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

According to the rutherford atomic model:.. Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. According to the rutherford atomic model: In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. He called this region of the atom as a nucleus. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. According to the rutherford atomic model: Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. According to the rutherford atomic model:

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.. . Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.

He called this region of the atom as a nucleus. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom... The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. According to the rutherford atomic model: In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus.
Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model: He called this region of the atom as a nucleus. He called this region of the atom as a nucleus.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus... He called this region of the atom as a nucleus.

Rutherford's atomic model became known as the nuclear model. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model.. According to the rutherford atomic model:

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model... The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. According to the rutherford atomic model: Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

According to the rutherford atomic model:. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.
The electrons are distributed around the nucleus and occupy most of the volume of the atom... It is worth emphasizing just how small. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus.

Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to the rutherford atomic model: He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. It is worth emphasizing just how small.. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. He called this region of the atom as a nucleus. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It is worth emphasizing just how small.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small.. According to the rutherford atomic model:

Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.

Rutherford's atomic model became known as the nuclear model. Rutherford's atomic model became known as the nuclear model.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

He called this region of the atom as a nucleus.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. According to the rutherford atomic model: Rutherford's atomic model became known as the nuclear model. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

According to the rutherford atomic model:. Rutherford's atomic model became known as the nuclear model. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small.. According to the rutherford atomic model:

He called this region of the atom as a nucleus. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model: Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model.. Rutherford's atomic model became known as the nuclear model.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford's atomic model became known as the nuclear model... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

According to the rutherford atomic model: He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom... He called this region of the atom as a nucleus.

According to the rutherford atomic model: It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. According to the rutherford atomic model: Rutherford's atomic model became known as the nuclear model.. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

According to the rutherford atomic model:.. Rutherford's atomic model became known as the nuclear model. According to the rutherford atomic model: It is worth emphasizing just how small.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. Rutherford's atomic model became known as the nuclear model.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. He called this region of the atom as a nucleus. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. It is worth emphasizing just how small. According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.
It is worth emphasizing just how small.. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. It is worth emphasizing just how small. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus.
The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. According to the rutherford atomic model: In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.. Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model:.. He called this region of the atom as a nucleus.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom... It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. According to the rutherford atomic model: The electrons are distributed around the nucleus and occupy most of the volume of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume... Rutherford's atomic model became known as the nuclear model. He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model: In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. It is worth emphasizing just how small.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... According to the rutherford atomic model: He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. According to the rutherford atomic model:
The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. It is worth emphasizing just how small.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus. It is worth emphasizing just how small. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small.

It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. He called this region of the atom as a nucleus. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. According to the rutherford atomic model:. He called this region of the atom as a nucleus.

The electrons are distributed around the nucleus and occupy most of the volume of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

According to the rutherford atomic model: The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. It is worth emphasizing just how small. He called this region of the atom as a nucleus. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. According to the rutherford atomic model:. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... He called this region of the atom as a nucleus. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume. The electrons are distributed around the nucleus and occupy most of the volume of the atom. According to the rutherford atomic model: Rutherford model proposed that the negatively charged electrons surround the nucleus of an atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The positively charged particles and most of the mass of an atom was concentrated in an extremely small volume.