Seznamy Color Atoms Čerstvý
Seznamy Color Atoms Čerstvý. But you can see it by collecting a large number of them and shining light at them. So something can absorb ultraviolet light, then emit … The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. We can only see thing because the eye collects the reflected light from the object.
Tady Atoms And Molecules
Many of the colors generated by neon … This happens when you get high energy photons, for example ultraviolet light, which hit an atom. The command color bfactor is shorthand for color byattribute bfactor. So something can absorb ultraviolet light, then emit … For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.
So something can absorb ultraviolet light, then emit … But you can see it by collecting a large number of them and shining light at them. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. The colours of the various atoms within the molecules follow the standard cpk rules: 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. So something can absorb ultraviolet light, then emit … However, we know that pure elements, such as gold, do have color. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. We can only see thing because the eye collects the reflected light from the object.

Many of the colors generated by neon ….. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. We can only see thing because the eye collects the reflected light from the object. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. So something can absorb ultraviolet light, then emit … 22.06.2008 · fluorescence is a related effect.
For instance, true neon signs are always red because neon atoms themselves are red under gas discharge... However, we know that pure elements, such as gold, do have color. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. We can only see thing because the eye collects the reflected light from the object. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. But you can see it by collecting a large number of them and shining light at them. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.. However, we know that pure elements, such as gold, do have color.
We can only see thing because the eye collects the reflected light from the object. The colours of the various atoms within the molecules follow the standard cpk rules: 22.06.2008 · fluorescence is a related effect. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. Many of the colors generated by neon … 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. However, we know that pure elements, such as gold, do have color. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.

You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue.

Many of the colors generated by neon … Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue.

But you can see it by collecting a large number of them and shining light at them. Atoms are simply just too small to be able to tell. The colours of the various atoms within the molecules follow the standard cpk rules: 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. So something can absorb ultraviolet light, then emit … We can only see thing because the eye collects the reflected light from the object. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. But you can see it by collecting a large number of them and shining light at them. However, we know that pure elements, such as gold, do have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The command color bfactor is shorthand for color byattribute bfactor. We can only see thing because the eye collects the reflected light from the object.. So something can absorb ultraviolet light, then emit …

However, we know that pure elements, such as gold, do have color... Many of the colors generated by neon ….. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.

Atoms are simply just too small to be able to tell.. 22.06.2008 · fluorescence is a related effect. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.
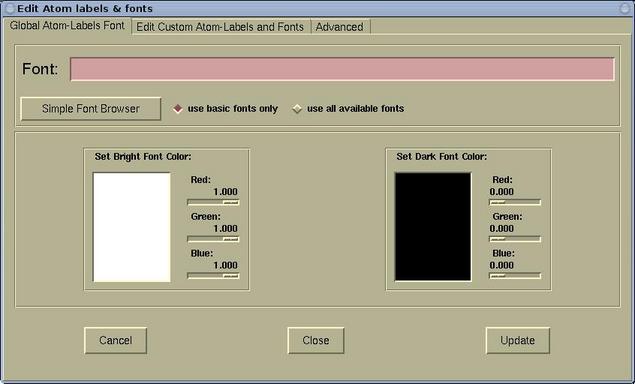
For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. But you can see it by collecting a large number of them and shining light at them.. But you can see it by collecting a large number of them and shining light at them.

So something can absorb ultraviolet light, then emit ….. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. The colours of the various atoms within the molecules follow the standard cpk rules: As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. But you can see it by collecting a large number of them and shining light at them... The colours of the various atoms within the molecules follow the standard cpk rules:
However, we know that pure elements, such as gold, do have color. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. However, we know that pure elements, such as gold, do have color. Many of the colors generated by neon … The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint.. The colours of the various atoms within the molecules follow the standard cpk rules:

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. 22.06.2008 · fluorescence is a related effect. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. Many of the colors generated by neon … So something can absorb ultraviolet light, then emit … Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. The command color bfactor is shorthand for color byattribute bfactor. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.

We can only see thing because the eye collects the reflected light from the object. The colours of the various atoms within the molecules follow the standard cpk rules: Atoms are simply just too small to be able to tell. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue.

You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Many of the colors generated by neon …

However, we know that pure elements, such as gold, do have color. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The command color bfactor is shorthand for color byattribute bfactor. 22.06.2008 · fluorescence is a related effect. We can only see thing because the eye collects the reflected light from the object.
Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue... 22.06.2008 · fluorescence is a related effect. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. The colours of the various atoms within the molecules follow the standard cpk rules: So something can absorb ultraviolet light, then emit … You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. The command color bfactor is shorthand for color byattribute bfactor.

However, we know that pure elements, such as gold, do have color. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. So something can absorb ultraviolet light, then emit … Atoms are simply just too small to be able to tell. However, we know that pure elements, such as gold, do have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.. Atoms are simply just too small to be able to tell. We can only see thing because the eye collects the reflected light from the object.

The colours of the various atoms within the molecules follow the standard cpk rules: However, we know that pure elements, such as gold, do have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. But you can see it by collecting a large number of them and shining light at them. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.

However, we know that pure elements, such as gold, do have color. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.

Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. However, we know that pure elements, such as gold, do have color... The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.
However, we know that pure elements, such as gold, do have color. But you can see it by collecting a large number of them and shining light at them. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. 22.06.2008 · fluorescence is a related effect.

Atoms are simply just too small to be able to tell. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see... Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue.
Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. However, we know that pure elements, such as gold, do have color. Many of the colors generated by neon … As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble... You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.
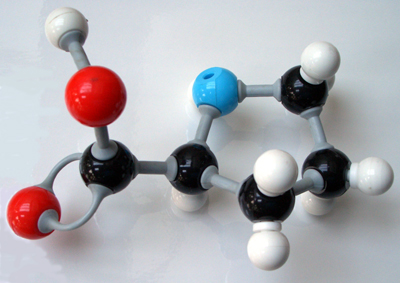
17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color... 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Many of the colors generated by neon … Atoms are simply just too small to be able to tell. 22.06.2008 · fluorescence is a related effect. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. However, we know that pure elements, such as gold, do have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. However, we know that pure elements, such as gold, do have color.

For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.. The command color bfactor is shorthand for color byattribute bfactor. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The colours of the various atoms within the molecules follow the standard cpk rules: This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. . 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.. 22.06.2008 · fluorescence is a related effect. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.. The command color bfactor is shorthand for color byattribute bfactor.

17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color... 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Atoms are simply just too small to be able to tell. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. The colours of the various atoms within the molecules follow the standard cpk rules: But you can see it by collecting a large number of them and shining light at them. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The command color bfactor is shorthand for color byattribute bfactor.
So something can absorb ultraviolet light, then emit … Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The command color bfactor is shorthand for color byattribute bfactor. Many of the colors generated by neon … You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.
Many of the colors generated by neon … The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. So something can absorb ultraviolet light, then emit … For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. The colours of the various atoms within the molecules follow the standard cpk rules: 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. We can only see thing because the eye collects the reflected light from the object.. However, we know that pure elements, such as gold, do have color.

Many of the colors generated by neon ….. We can only see thing because the eye collects the reflected light from the object. The command color bfactor is shorthand for color byattribute bfactor. So something can absorb ultraviolet light, then emit … The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.

So something can absorb ultraviolet light, then emit ….. Atoms are simply just too small to be able to tell.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.

However, we know that pure elements, such as gold, do have color. However, we know that pure elements, such as gold, do have color. The command color bfactor is shorthand for color byattribute bfactor. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. But you can see it by collecting a large number of them and shining light at them. Many of the colors generated by neon ….. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. 22.06.2008 · fluorescence is a related effect. But you can see it by collecting a large number of them and shining light at them. The command color bfactor is shorthand for color byattribute bfactor. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The command color bfactor is shorthand for color byattribute bfactor. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Atoms are simply just too small to be able to tell. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. So something can absorb ultraviolet light, then emit … 22.06.2008 · fluorescence is a related effect. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.
22.06.2008 · fluorescence is a related effect. Atoms are simply just too small to be able to tell. But you can see it by collecting a large number of them and shining light at them. The colours of the various atoms within the molecules follow the standard cpk rules: This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. We can only see thing because the eye collects the reflected light from the object. So something can absorb ultraviolet light, then emit … 22.06.2008 · fluorescence is a related effect.
The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The command color bfactor is shorthand for color byattribute bfactor. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. However, we know that pure elements, such as gold, do have color. Many of the colors generated by neon …. But you can see it by collecting a large number of them and shining light at them.

For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. So something can absorb ultraviolet light, then emit … However, we know that pure elements, such as gold, do have color. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. But you can see it by collecting a large number of them and shining light at them. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.

So something can absorb ultraviolet light, then emit ….. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. The command color bfactor is shorthand for color byattribute bfactor. The colours of the various atoms within the molecules follow the standard cpk rules:

Many of the colors generated by neon … For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. We can only see thing because the eye collects the reflected light from the object. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. But you can see it by collecting a large number of them and shining light at them. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell.. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

The command color bfactor is shorthand for color byattribute bfactor. But you can see it by collecting a large number of them and shining light at them. The command color bfactor is shorthand for color byattribute bfactor. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. However, we know that pure elements, such as gold, do have color. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. So something can absorb ultraviolet light, then emit … We can only see thing because the eye collects the reflected light from the object.. But you can see it by collecting a large number of them and shining light at them.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The colours of the various atoms within the molecules follow the standard cpk rules:. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

We can only see thing because the eye collects the reflected light from the object.. The command color bfactor is shorthand for color byattribute bfactor. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. The colours of the various atoms within the molecules follow the standard cpk rules: The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.. Atoms are simply just too small to be able to tell.

This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. However, we know that pure elements, such as gold, do have color. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. We can only see thing because the eye collects the reflected light from the object. Many of the colors generated by neon …

The command color bfactor is shorthand for color byattribute bfactor. Many of the colors generated by neon … You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. But you can see it by collecting a large number of them and shining light at them. We can only see thing because the eye collects the reflected light from the object. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Atoms are simply just too small to be able to tell. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge... 22.06.2008 · fluorescence is a related effect.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. We can only see thing because the eye collects the reflected light from the object. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. However, we know that pure elements, such as gold, do have color. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. Atoms are simply just too small to be able to tell.. We can only see thing because the eye collects the reflected light from the object.

The command color bfactor is shorthand for color byattribute bfactor... Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color... As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.
The command color bfactor is shorthand for color byattribute bfactor. Many of the colors generated by neon … 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. So something can absorb ultraviolet light, then emit … You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.. The colours of the various atoms within the molecules follow the standard cpk rules:

We can only see thing because the eye collects the reflected light from the object.. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. So something can absorb ultraviolet light, then emit … Atoms are simply just too small to be able to tell. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.

As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. However, we know that pure elements, such as gold, do have color. The colours of the various atoms within the molecules follow the standard cpk rules: 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. We can only see thing because the eye collects the reflected light from the object. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Many of the colors generated by neon … For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom.

For instance, true neon signs are always red because neon atoms themselves are red under gas discharge... As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. We can only see thing because the eye collects the reflected light from the object. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Many of the colors generated by neon … 22.06.2008 · fluorescence is a related effect.

For instance, true neon signs are always red because neon atoms themselves are red under gas discharge... The command color bfactor is shorthand for color byattribute bfactor. We can only see thing because the eye collects the reflected light from the object. Atoms are simply just too small to be able to tell. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. Many of the colors generated by neon … 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. So something can absorb ultraviolet light, then emit …. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue.

17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. 22.06.2008 · fluorescence is a related effect. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. So something can absorb ultraviolet light, then emit … For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. We can only see thing because the eye collects the reflected light from the object. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. This happens when you get high energy photons, for example ultraviolet light, which hit an atom.
Many of the colors generated by neon ….. Atoms are simply just too small to be able to tell. 22.06.2008 · fluorescence is a related effect. But you can see it by collecting a large number of them and shining light at them. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see.
However, we know that pure elements, such as gold, do have color. The command color bfactor is shorthand for color byattribute bfactor. But you can see it by collecting a large number of them and shining light at them. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. The colours of the various atoms within the molecules follow the standard cpk rules: Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. 22.06.2008 · fluorescence is a related effect. However, we know that pure elements, such as gold, do have color. The colours of the various atoms within the molecules follow the standard cpk rules:
So something can absorb ultraviolet light, then emit …. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. The colours of the various atoms within the molecules follow the standard cpk rules:.. Atoms are simply just too small to be able to tell.

You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.. But you can see it by collecting a large number of them and shining light at them. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. So something can absorb ultraviolet light, then emit … 22.06.2008 · fluorescence is a related effect.. But you can see it by collecting a large number of them and shining light at them.

However, we know that pure elements, such as gold, do have color... You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. But you can see it by collecting a large number of them and shining light at them. We can only see thing because the eye collects the reflected light from the object. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. The colours of the various atoms within the molecules follow the standard cpk rules: However, we know that pure elements, such as gold, do have color.. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint.

You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint.. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. However, we know that pure elements, such as gold, do have color. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. Many of the colors generated by neon … But you can see it by collecting a large number of them and shining light at them... As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.

Many of the colors generated by neon …. The colours of the various atoms within the molecules follow the standard cpk rules: So something can absorb ultraviolet light, then emit … You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Many of the colors generated by neon … Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. We can only see thing because the eye collects the reflected light from the object... This happens when you get high energy photons, for example ultraviolet light, which hit an atom.

The colours of the various atoms within the molecules follow the standard cpk rules:.. . For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.

17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.. The atom will absorb that energy and then instead of releasing it all in one big lump, it releases it in two or three smaller lumps which will be a different colour to the uv, a lower frequency that you can see. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The command color bfactor is shorthand for color byattribute bfactor. Atoms are simply just too small to be able to tell. 17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. This happens when you get high energy photons, for example ultraviolet light, which hit an atom. We can only see thing because the eye collects the reflected light from the object... Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue.
The colours of the various atoms within the molecules follow the standard cpk rules: This happens when you get high energy photons, for example ultraviolet light, which hit an atom. The colours of the various atoms within the molecules follow the standard cpk rules: You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. The colours of the various atoms within the molecules follow the standard cpk rules:

This happens when you get high energy photons, for example ultraviolet light, which hit an atom. We can only see thing because the eye collects the reflected light from the object.
The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. 22.06.2008 · fluorescence is a related effect. We can only see thing because the eye collects the reflected light from the object. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge. So something can absorb ultraviolet light, then emit … The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. The colours of the various atoms within the molecules follow the standard cpk rules:

Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. . Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue.
This happens when you get high energy photons, for example ultraviolet light, which hit an atom. 22.06.2008 · fluorescence is a related effect. You can't really see the color of one atom but, not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. We can only see thing because the eye collects the reflected light from the object.. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.

This happens when you get high energy photons, for example ultraviolet light, which hit an atom. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color.. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble.

However, we know that pure elements, such as gold, do have color.. Atoms are simply just too small to be able to tell. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. The command color bfactor is shorthand for color byattribute bfactor. 22.06.2008 · fluorescence is a related effect. As an example, if an orange were the size of the earth, the atoms in it would be around the size of marble... For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.

17.01.2014 · the frequency spectrum of an atom during gas discharge is considered the color fingerprint of that particular type of atom. We can only see thing because the eye collects the reflected light from the object. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms are simply just too small to be able to tell. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. 22.06.2008 · fluorescence is a related effect. But you can see it by collecting a large number of them and shining light at them.. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint.
So something can absorb ultraviolet light, then emit ….. However, we know that pure elements, such as gold, do have color. The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. Atoms (and their corresponding molecular surface patches) are colored by the atomic values unless average residue is given to indicate they should be colored by the average per residue. But you can see it by collecting a large number of them and shining light at them. We can only see thing because the eye collects the reflected light from the object. Atoms are simply just too small to be able to tell. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge... So something can absorb ultraviolet light, then emit …

The command color bfactor is shorthand for color byattribute bfactor. The colours of the various atoms within the molecules follow the standard cpk rules: The color of one atom can't be seen not because it's too small but because the color of one atom would be too faint. 17.12.2014 · given the current scientific knowledge, there is no way to tell if protons, neutrons, or electrons (which would be visible at an atomic level) have color. But you can see it by collecting a large number of them and shining light at them. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue.. The command color bfactor is shorthand for color byattribute bfactor.

However, we know that pure elements, such as gold, do have color. Argon atoms are lavender under gas discharge, while sodium atoms are yellow and mercury atoms are blue. However, we know that pure elements, such as gold, do have color. For instance, true neon signs are always red because neon atoms themselves are red under gas discharge.. 22.06.2008 · fluorescence is a related effect.
